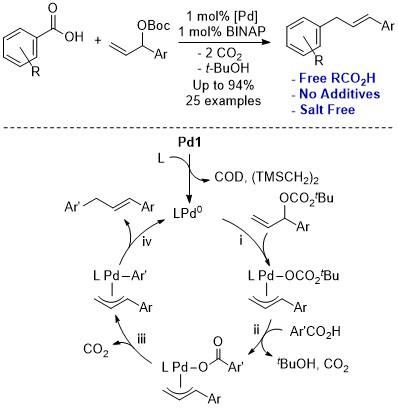
Densely and differentially functionalized aromatic and heteroaromatic compounds represent the core of numerous molecules of societal value including dyes, pharmaceuticals, flavors, fragrances, agrochemicals, materials, and catalyst ligands. The synthesis of complex (hetero)aromatics has been a central goal of organic synthesis since Perkin’s original isolation of mauveine dyes. Since Perkin’s time, numerous synthetic methods have been developed for the functionalization of (hetero)aromatics. Many populate the pages of introductory organic chemistry texts (e.g. EAS or SNAr). However, there are many unsolved problems for efficient (hetero)arene functionalization. If a native or ubiquitous functional group, such as a benzoic acid, could be used in (hetero)arene functionalization it would offer tremendous advantages in efficiency and site specificity. Carboxylic acids are more stable, more readily available, and cheaper than boronic acids. Our lab is developing decarboxylative methods for the efficient synthesis of functionalized arenes.